【Nature immunology】
2025年10-12月论文导读
期刊介绍:
Nature Immunology是Nature杂志的免疫学分册,创刊于2000年,也是该领域经由同行评审的权威科学期刊。该杂志由NATURE RESEARCH出版集团按每月一期出版。该期刊是一本以医学-免疫学综合研究为特色的国际期刊,其涵盖的领域包括但不限于先天免疫和炎症,发育,免疫受体,信号传导和凋亡,抗原呈递,基因调控和重组,细胞和全身免疫,疫苗,免疫耐受,自身免疫,肿瘤免疫学和微生物免疫病理学。最新影响因子指数为28.3。
本期文献导读将呈现2025年10月至2025年12月三个月内的主要刊物内容。

Volume 26, Issue 10
本期共发表论著13篇。
1. Delaying pyroptosis with an AI-screened gasdermin D pore blocker mitigates inflammatory response
使用AI筛选的气孔素D孔道阻滞剂延缓细胞焦亡可减轻炎症反应

第三军医大学大坪医院、昆明医科大学附属延安医院、中国科学院成都生物研究所及西南大学
The formation of membrane pores by cleaved N-terminal gasdermin D (GSDMD-NT) results in the release of cytokines and inflammatory cell death, known as pyroptosis. Blocking GSDMD-NT pores is an attractive and promising strategy for mitigating inflammation. Here we demonstrate that SK56, an artificial intelligence-screened peptide, effectively obstructs GSDMD-NT pores and inhibits pyroptosis and cytokine release in macrophages and human peripheral blood leukocyte-induced pyroptosis. SK56 prevents septic death induced by lipopolysaccharide or cecal ligation and puncture surgery in mice. SK56 does not influence cleavage of interleukin-1β or GSDMD. Instead, SK56 inhibits the release of cytokines from pyroptotic macrophages, mitigates the activation of primary mouse dendritic cells triggered by incubation with pyroptotic cytomembranes and prevents widespread cell death of human alveolar organoids in an organoid–macrophage coculture model. SK56 blocks GSDMD-NT pores on lipid-bilayer nanoparticles and enters pyroptotic macrophages to inhibit mitochondrial damage. SK56 presents new therapeutic possibilities for counteracting inflammation, which is implicated in numerous diseases.
被切割的N端气孔素D(GSDMD-NT)形成的膜孔会导致细胞因子释放和炎症性细胞死亡,即细胞焦亡。阻断GSDMD-NT孔道是一种极具吸引力且前景广阔的抑制炎症策略。文章中,研究者证明人工智能筛选的肽段SK56能有效堵塞GSDMD-NT孔道,并在巨噬细胞及人外周血白细胞诱导的焦亡模型中抑制细胞焦亡和细胞因子释放。SK56能预防由脂多糖或盲肠结扎穿孔手术引发的小鼠脓毒症死亡。SK56不影响白细胞介素-1β或GSDMD的切割过程,而是通过抑制焦亡巨噬细胞的细胞因子释放、减轻由焦亡细胞膜触发的原代小鼠树突状细胞活化,并在类器官-巨噬细胞共培养模型中阻止人肺泡类器官的广泛细胞死亡。SK56能阻断脂质双分子层纳米颗粒上的GSDMD-NT孔道,并进入发生焦亡的巨噬细胞以抑制线粒体损伤。SK56为对抗涉及多种疾病的炎症反应提供了新的治疗可能性。
2. Type A cholesterol-dependent cytolysins translocate to the trans-Golgi network for NLRP3 inflammasome activation
A型胆固醇依赖性溶细胞素易位至高尔基体反式网络,从而激活NLRP3炎症小体

芝加哥大学微生物学系
Cholesterol-dependent cytolysins (CDCs) constitute the largest group of pore-forming toxins and serve as critical virulence factors for diverse pathogenic bacteria. Several CDCs are known to activate the NLRP3 inflammasome, although the mechanisms are unclear. Here we discovered that multiple CDCs, which we referred to as type A CDCs, were internalized and translocated to the trans-Golgi network (TGN) to remodel it into a platform for NLRP3 activation through a unique peeling membrane mechanism. Potassium efflux was dispensable for CDC-mediated TGN remodeling and NLRP3 recruitment, but was required for the recruitment of the downstream adaptor ASC. In contrast, desulfolysin, which we referred to as type B CDC, was not internalized or translocated to the TGN due to its distinct C-terminal domain 4, despite potent pore formation on the plasma membrane, and hence could not activate NLRP3. Our discoveries uncovered the ability of CDCs to directly remodel an intracellular organelle for inflammatory response.
胆固醇依赖性溶细胞素(CDCs)是最大的一类成孔毒素,也是多种致病菌的关键毒力因子。目前已知数种CDCs能够激活NLRP3炎症小体,但其作用机制尚不明确。该研究发现,多种被归类为A型CDCs的毒素可通过内化作用转运至高尔基体反式网络(TGN),并借助独特的膜剥离机制将TGN重塑为激活NLRP3的平台。钾离子外流对于CDC介导的TGN重塑和NLRP3募集并非必需,但对下游接头蛋白ASC的募集不可或缺。与之相对,被归类为B型CDC的脱硫溶素,尽管能在质膜上形成强效孔道,却因其独特的C端结构域4而无法内化或转运至TGN,因而不能激活NLRP3。该研究发现揭示了CDCs直接重塑细胞内细胞器以触发炎症反应的能力。
3. Epigenetic imprinting in innate lymphoid cell precursors directs the lineage segregation of innate lymphoid cells
先天淋巴细胞前体中的表观遗传印记调控先天淋巴细胞的谱系定向分化

中国科学院微生物研究所、厦门大学医学院附属心血管病医院、中国农业大学动物科学技术学院及北京大学基础医学院
Innate lymphoid cells (ILCs) are essential for mucosal homeostasis, but the epigenetic regulation of their lineage segregation remains elusive. Here we simultaneously profiled the single-cell DNA methylome, chromatin accessibility and transcriptome of ILC subsets and ILC precursors (ILCPs) and found that ILCPs could be divided into two subgroups (ILCP1 and ILCP2). ILCP2s had highly heterogeneous DNA methylation profiles and could be divided into three groups according to their DNA methylation characteristics, which matched those of ILC subsets. We identified the signature methylation regions (SMRs) of each ILC subset and traced the DNA methylation imprinting during ILCP differentiation. ILCP2s with hypomethylated SMRs characteristic of ILC subsets differentiated into those subsets. DNA methylation editing of SMRs suppressed ILC lineage segregation, while deletion of Dnmt1 in ILCPs abrogated the heterogeneous distribution of SMRs and resulted in ILC differentiation defects. These findings provide evidence that epigenetic imprinting determines lineage segregation during immune cell development.
先天淋巴细胞(ILCs)对维持黏膜稳态至关重要,但其谱系分化的表观遗传调控机制仍不明确。该研究通过同步分析ILC亚群及ILC前体细胞(ILCPs)的单细胞DNA甲基化组、染色质可及性与转录组,发现ILCPs可划分为两个亚群(ILCP1和ILCP2)。其中ILCP2亚群呈现高度异质性的DNA甲基化谱,根据其甲基化特征可进一步分为三组,且这些特征与成熟ILC亚群相匹配。研究鉴定了各ILC亚群的特征性甲基化区域(SMRs),并追踪了ILCP分化过程中的DNA甲基化印记动态。研究发现,具有特定ILC亚群低甲基化SMR特征的ILCP2细胞会向对应亚群分化。通过编辑SMRs的DNA甲基化状态可抑制ILC谱系分化,而在ILCPs中敲除Dnmt1则破坏了SMRs的异质性分布,导致ILC分化缺陷。这些发现为表观遗传印记决定免疫细胞发育过程中的谱系分化提供了直接证据。
4. Damage-induced IL-18 stimulates thymic NK cells limiting endogenous tissue regeneration
损伤诱导的白细胞介素-18通过刺激胸腺自然杀伤细胞抑制内源性组织再生

华盛顿大学医学院、弗雷德·哈钦森癌症中心
Interleukin-18 (IL-18) is an acute-phase proinflammatory molecule crucial for mediating viral clearance by activating T helper 1 CD4+ T cells, cytotoxic CD8+ T cells and natural killer (NK) cells. Here, we show that mature IL-18 is generated in the thymus following numerous distinct forms of tissue damage, all of which cause caspase-1-mediated immunogenic cell death. We report that IL-18-stimulated cytotoxic NK cells limit endogenous thymic regeneration, a critical process that ensures the restoration of immune competence after acute insults such as stress, infection, chemotherapy and radiation. NK cells suppress thymus recovery by aberrantly targeting thymic epithelial cells, which act as the master regulators of organ function and regeneration. Together, our data reveal a new pathway regulating tissue regeneration in the thymus and suggest IL-18 as a potential therapeutic target to boost thymic function. Moreover, given the enthusiasm for IL-18 as a cancer immunotherapy due to its capacity to elicit a type 1 immune response, these findings also offer insight into potential off-target effects.
白细胞介素-18(IL-18)是一种急性期促炎分子,通过激活Th1 CD4⁺ T细胞、细胞毒性CD8⁺ T细胞和自然杀伤细胞(NK细胞)在病毒清除过程中发挥关键作用。该研究发现,在多种不同形式的组织损伤后,成熟的IL-18会在胸腺内生成,且这些损伤均会引发caspase-1介导的免疫原性细胞死亡。研究表明,IL-18激活的细胞毒性NK细胞会抑制内源性胸腺再生——这一关键过程对于机体在应激、感染、化疗及辐射等急性损伤后恢复免疫功能至关重要。NK细胞通过异常攻击胸腺上皮细胞来抑制胸腺修复,而胸腺上皮细胞正是胸腺器官功能与再生的核心调控者。综上,研究揭示了一条调控胸腺组织再生的新通路,并提示IL-18可能成为增强胸腺功能的潜在治疗靶点。此外,鉴于IL-18因其诱导1型免疫应答的能力而被视为癌症免疫疗法的热点分子,这些发现也为理解其潜在脱靶效应提供了重要参考。
5. Pre-TCR-targeted immunotherapy for T cell acute lymphoblastic leukemia
靶向前T细胞受体的T细胞急性淋巴细胞白血病免疫疗法

塞韦罗·奥乔亚分子生物学中心
Targeted immunotherapy for T cell acute lymphoblastic leukemia (T-ALL), an aggressive tumor of developing T cell progenitors, is an urgent unmet need, especially for relapsed/refractory disease. Selective T-ALL targeting is challenging due to the shared antigen expression between leukemic and normal T cells. Here we identify the pre-T cell receptor (pre-TCR), a surface receptor essential for T cell development, as a biomarker of leukemia-initiating cells (LICs) in human T-ALL. Loss-of-function genetic approaches demonstrate that pre-TCR signaling is necessary for LIC activity and tumor progression in pre-TCR+ T-ALL patient xenografts in mice. Furthermore, we demonstrate the specific therapeutic targeting of the pre-TCR with a monoclonal antibody against the invariant pTα subunit of the human pre-TCR, and validate an anti-pTα antibody–drug conjugate in vivo treatment as a potent immunotherapy for inhibiting LIC activity and tumor progression of T-ALL in mice. These findings reveal the suitability of pre-TCR targeting as a promising therapy for the treatment of individuals with relapsed/refractory T-ALL expressing the pre-TCR.
T细胞急性淋巴细胞白血病(T-ALL)是一种起源于发育中T细胞前体的侵袭性肿瘤,其靶向免疫治疗——尤其是针对复发/难治性疾病——是目前亟待满足的临床需求。由于白血病细胞与正常T细胞存在共有的抗原表达,实现选择性靶向T-ALL面临巨大挑战。该研究发现,前T细胞受体(pre-TCR)——一种对T细胞发育至关重要的表面受体——可作为人类T-ALL中白血病起始细胞(LICs)的生物标志物。通过功能缺失性遗传学方法,研究证实pre-TCR信号传导对于pre-TCR阳性T-ALL患者来源的小鼠异种移植模型中LIC的活性及肿瘤进展是必需的。进一步,研究人员利用靶向人pre-TCR恒定链pTα亚基的单克隆抗体实现了对pre-TCR的特异性治疗性靶向,并在体内验证了抗pTα抗体-药物偶联物作为有效免疫疗法,能够抑制T-ALL小鼠模型中LIC活性及肿瘤进展。这些发现揭示了靶向pre-TCR作为治疗表达pre-TCR的复发/难治性T-ALL患者的潜力。
6. TCR-engineered T cells targeting a shared β-catenin mutation eradicate solid tumors
靶向β-连环蛋白共有突变的TCR工程化T细胞可根除实体肿瘤

奥斯陆大学医院癌症免疫学系
HLA-bound peptides encoded by recurrent driver mutations are candidate targets for T cell-directed immunotherapy. Here we identify two neopeptides encoded by the CTNNB1S37F mutation presented on the frequent HLA-A*02:01 and HLA-A*24:02 molecules in cell lines naturally expressing the mutation and HLA alleles. This mutation leads to a gain of function in β-catenin and is estimated to occur in >7,000 new cancer cases annually in the United States. T cell receptors (TCRs) that specifically recognize the mutant peptides were isolated from naive healthy donor T cells. T cells redirected with CTNNB1-S37F TCRs efficiently killed CTNNB1S37F+ cell lines and patient-derived organoids in vitro and eradicated established tumors in a melanoma cell line mouse model and a patient-derived xenograft model of endometrial adenocarcinoma naturally expressing the mutation and the restricting HLA. We propose that TCR-T cells targeting CTNNB1-S37F can serve as a basis for solid cancer immunotherapy.
由复发性驱动突变编码的HLA结合肽是T细胞导向免疫疗法的潜在靶点。该研究发现,在天然表达该突变及对应HLA等位基因的细胞系中,CTNNB1S37F突变编码的两个新肽可被高频率的HLA-A*02:01和HLA-A*24:02分子递呈。该突变导致β-连环蛋白功能增强,据估计在美国每年新增超过7,000例相关癌症病例。研究人员从健康供体的初始T细胞中分离出能特异性识别这些突变肽的T细胞受体(TCR)。经CTNNB1-S37F特异性TCR改造的T细胞,在体外能有效杀伤CTNNB1S37F阳性细胞系及患者来源类器官,并在天然表达该突变及限制性HLA的黑色素瘤细胞系小鼠模型和子宫内膜腺癌患者来源异种移植模型中根除已形成的肿瘤。研究认为,靶向CTNNB1-S37F的TCR-T细胞可作为实体瘤免疫疗法的开发基础。
7. SATB1 is a key regulator of quiescence in stem-like CD8+ T cells
SATB1是调控干细胞样CD8⁺ T细胞静息状态的关键因子

西北大学范伯格医学院、威斯康星医学院、上海交通大学医学院及加州大学旧金山分校
Stem-like progenitor CD8+ T (TPRO) cells sustain cytotoxic immunity during chronic infection and cancer through quiescence, multipotency and self-renewal, hallmarks shared with memory T cells. However, how these properties are maintained under persistent antigen stimulation remains unclear. Here we identify the genomic organizer SATB1 as selectively enriched in both TPRO and memory CD8+ T cells. Given its role in promoting quiescence in hematopoietic stem cells, we hypothesized that SATB1 supports CD8+ T cell stemness. Using CD8+ T cell-specific CRISPR deletion of the Satb1 gene, we show that SATB1 is essential for maintaining TPRO cells during chronic lymphocytic choriomeningitis virus infection and for memory CD8+ T cell formation during acute infection. Multi-omic profiling revealed that SATB1 regulates the chromatin accessibility, transcriptional activity and genome architecture of stemness-associated genes including Tcf7, Bach2 and Myb. These findings reveal a critical role for SATB1 in preserving the transcriptional and epigenetic programs that sustain the stem-like state of antigen-specific CD8+ T cells.
干细胞样前体CD8⁺ T细胞(TPRO细胞)通过静息状态、多能性和自我更新能力维持慢性感染和癌症期间的细胞毒性免疫,这些特性与记忆性T细胞共有。然而,在持续抗原刺激下这些特性如何维持仍不清楚。该研究发现基因组组织因子SATB1在TPRO细胞和记忆性CD8⁺ T细胞中均特异性富集。鉴于SATB1在促进造血干细胞静息状态中已知的作用,研究假设其可能支持CD8⁺ T细胞的干性。通过CD8⁺ T细胞特异性CRISPR敲除Satb1基因,研究发现SATB1对于慢性淋巴细胞性脉络丛脑膜炎病毒感染期间TPRO细胞的维持以及急性感染中记忆性CD8⁺ T细胞的形成至关重要。多组学分析揭示,SATB1通过调节染色质可及性、转录活性和基因组三维结构,调控包括Tcf7、Bach2和Myb在内的干性相关基因。这些发现明确了SATB1在维持抗原特异性CD8⁺ T细胞干细胞样状态的转录与表观遗传程序中发挥关键作用。
8. Lymph nodes fuel KLF2-dependent effector CD8+ T cell differentiation during chronic infection and checkpoint blockade
慢性感染与检查点阻断期间淋巴结驱动KLF2依赖性效应CD8⁺ T细胞分化

墨尔本大学微生物学与免疫学系、波恩大学医院
Exhausted CD8+ T (TEX) cell responses are maintained by precursors of exhausted T (TPEX) cells that possess high self-renewal and developmental potential. TPEX cells also drive the proliferative burst of effector T cells upon therapeutic immune checkpoint blockade (ICB). However, the spatial context and signals that regulate their differentiation and function are not well defined. Here we identify developmental and functional compartmentalization of TPEX and TEX cells across secondary lymphoid organs during chronic infection. We show that stem-like CD62L+ TPEX and effector-like CX3CR1+ TEX cells constitute a distinct developmental lineage that is promoted by the lymph node (LN) microenvironment and dependent on the transcription factor KLF2. LNs act as a niche in which migratory dendritic cells provide antigen and costimulatory signals to maintain the proliferative fitness of CD62L+ TPEX cells and generation of CX3CR1+ TEX cells. Moreover, LNs exclusively drive the proliferative burst and systemic dissemination of CX3CR1+ TEX cells during ICB. Thus, our findings identify a unique role for LNs in the maintenance of T cell differentiation and function during systemic chronic infection and ICB therapy.
耗竭性CD8⁺ T细胞(TEX细胞)应答由其具有高度自我更新和发育潜能的耗竭前体T细胞(TPEX细胞)维持。TPEX细胞还能在治疗性免疫检查点阻断(ICB)时驱动效应T细胞的增殖爆发。然而,调控其分化与功能的空间环境及信号机制尚不明确。该研究发现在慢性感染期间,TPEX细胞和TEX细胞在次级淋巴器官中存在发育与功能的空间区隔。研究表明,干细胞样CD62L⁺ TPEX细胞与效应细胞样CX3CR1⁺ TEX细胞构成一个独特的发育谱系,该谱系受淋巴结(LN)微环境促进并依赖转录因子KLF2。淋巴结作为生态位,其中迁移性树突状细胞通过提供抗原和共刺激信号来维持CD62L⁺ TPEX细胞的增殖适应性并促进CX3CR1⁺ TEX细胞的生成。更重要的是,在ICB治疗期间,淋巴结专一性地驱动CX3CR1⁺ TEX细胞的增殖爆发和全身性播散。因此,研究揭示了淋巴结在系统性慢性感染和ICB治疗期间维持T细胞分化与功能的独特作用。
9. Simultaneous STING and lymphotoxin-β receptor activation induces B cell responses in tertiary lymphoid structures to potentiate antitumor immunity
同步激活STING与淋巴毒素-β受体可诱导三级淋巴结构中B细胞应答,从而增强抗肿瘤免疫

约翰斯·霍普金斯奥驰儿童医院
B cell-rich tertiary lymphoid structures (TLS) are associated with favorable prognosis and positive response to immunotherapy in cancer. Here we show that simultaneous activation of innate immune effectors, STING and lymphotoxin-β receptor (LTβR), results in CD8+ T cell-dependent tumor suppression while inducing high endothelial venule development and germinal center-like B cell responses in tumors to generate functional TLS in a T cell-dependent manner. In a neoadjuvant setting, activation of STING and LTβR by their agonists effectively immunized mice against tumor recurrence, leading to long-term survival. STING activation alone was insufficient for inducing B cell-containing TLS or eliciting long-term therapeutic effects. However, when combined with LTβR activation, it improved the fitness of TLS with B cell expansion and maturation to IgG-producing long-lived plasma cells and memory cells, increased CD4+ T cell recruitment and memory CD8+ T cell expansion, and shifted the TH2/TH17 balance, resulting in the potentiation of humoral and cellular immunity against tumors. These findings suggest a therapeutic approach of simultaneously activating STING and lymphotoxin pathways.
富含B细胞的三级淋巴结构(TLS)与癌症患者的良好预后及对免疫治疗的积极反应相关。该研究发现,同时激活固有免疫效应分子STING和淋巴毒素-β受体(LTβR)可在依赖CD8⁺ T细胞的情况下抑制肿瘤,同时诱导肿瘤内高内皮微静脉发育和生发中心样B细胞应答,从而以T细胞依赖的方式形成功能性TLS。在新辅助治疗场景中,通过激动剂激活STING和LTβR能有效使小鼠产生抗肿瘤复发免疫,实现长期生存。单独激活STING不足以诱导含B细胞的TLS形成或引发长期治疗效果,但当与LTβR激活联合时,能通过促进B细胞扩增及分化为产IgG的长寿浆细胞和记忆细胞来改善TLS的功能状态,同时增加CD4⁺ T细胞募集和记忆性CD8⁺ T细胞扩增,并改变TH2/TH17平衡,最终协同增强抗肿瘤的体液免疫与细胞免疫。这些发现提示同时激活STING和淋巴毒素信号通路可能成为一种治疗策略。
10. Distinct roles for B cell-derived LTα3 and LTα1β2 in TNF-mediated ileitis
B细胞来源的LTα3与LTα1β2在TNF介导的回肠炎中具有不同的功能

华盛顿大学医学院病理学与免疫学系
Crohn’s disease pathology is modeled in TNFΔARE+/− mice that overproduce tumor necrosis factor (TNF) to drive disease through TNF receptors. An alternative ligand for TNF receptors, soluble LTα3, is produced by B cells, but has received scarce attention because LTα also partners with LTβ to generate membrane-tethered LTαβ2 that promotes tertiary lymphoid tissue—another feature of Crohn’s disease. We hypothesized that B cell-derived LTαβ2 would critically affect ileitis in TNFΔARE+/− mice. However, whereas deleting LTβ in B cells was essential for tertiary lymphoid tissue, disease pathology was minimally affected. By contrast, loss of B cell-derived LTα increased intestinal permeability, shrunk the pool of IgA+ ileal plasma cells, elevated cytokines and prompted weight loss, including loss of muscle mass—a systemic feature of Crohn’s disease. Neutralizing antibodies to LTα3 strongly augmented the cachexic-like effects of TNF. Thus, B cell-produced LTαβ2 and LTα3 have distinct roles in ileitis, with the role of LTα3 unexpectedly protective through counterbalancing TNF.
克罗恩病的病理特征在TNFΔARE+/−小鼠中得以模拟,该模型通过过度产生肿瘤坏死因子(TNF)并经由TNF受体驱动疾病。TNF受体的另一种配体——可溶性LTα3由B细胞产生,但因其常与LTβ结合形成膜锚定型LTαβ2以促进三级淋巴组织形成(克罗恩病的另一特征),所以长期以来未受足够重视。研究人员假设B细胞来源的LTαβ2会对TNFΔARE+/− 小鼠的回肠炎产生关键影响。然而,尽管在B细胞中敲除LTβ对于三级淋巴组织的形成至关重要,但对疾病病理的影响却微乎其微。与之形成对比的是,缺失B细胞来源的LTα会导致肠道通透性增加、回肠IgA⁺浆细胞池萎缩、细胞因子水平升高并引发体重减轻(包括肌肉流失)——这正是克罗恩病的全身性特征。使用LTα3 中和抗体显著增强了TNF引起的恶病质样效应。因此,B细胞产生的LTαβ2和 LTα3 在回肠炎中发挥着不同作用,其中 LTα3 出乎意料地通过拮抗TNF发挥保护作用。
11. A spatial map of MAPK-activated immunosuppressive myeloid populations in pediatric low-grade glioma
儿童低级别胶质瘤中MAPK活化免疫抑制性髓系细胞的空间分布图谱

加拿大卡尔加里大学、麦吉尔大学
Pediatric low-grade gliomas (pLGGs) are mitogen-activated protein kinase (MAPK) pathway-activated brain tumors prevalent in children and are associated with morbidity despite favorable survival. Here using imaging mass cytometry, we spatially characterized at the single-cell level the tumor microenvironment (TME) of 120 pLGG cases, considering age, molecular drivers, brain location and tumor subtype. Our analysis identified myeloid cells—including resident microglia and bone marrow-derived macrophages—as the predominant immune population in the TME, particularly in optic pathway tumors. Additionally, we discovered an immune signature predictive of progression-free survival. Spatial analysis identified specific cellular interactions, notably myeloid–myeloid contacts and macrophage-enriched regions harboring MAPK-activated, TIM-3+ myeloid cells, suggesting an immunosuppressive TME. Our study provides a comprehensive resource on the immune landscape of these pLGGs and underscores the immunosuppressive role of diverse myeloid infiltrates. These findings also indicate that combining TIM-3 blockade with MAPK inhibition might be a promising therapeutic strategy to target both the TME and oncogenic MAPK activation in pLGG tumors.
儿童低级别胶质瘤(pLGGs)是由丝裂原活化蛋白激酶(MAPK)通路异常激活的脑肿瘤,在儿童中高发,尽管生存率尚可但仍伴有显著并发症。该研究采用成像质谱流式技术,从单细胞水平对120例pLGG样本的肿瘤微环境(TME)进行了空间特征分析,并综合考虑了年龄、分子驱动因素、脑区定位及肿瘤亚型等变量。分析发现髓系细胞(包括常驻小胶质细胞和骨髓来源巨噬细胞)是TME中的主要免疫群体,尤其在视路肿瘤中更为突出。此外,研究鉴定出一个可预测无进展生存期的免疫特征谱。空间分析揭示了特定的细胞间相互作用,特别是髓系-髓系细胞直接接触以及富含MAPK活化、TIM-3⁺髓系细胞的巨噬细胞富集区域,这些发现提示了TME的免疫抑制特性。该研究为pLGG的免疫图谱提供了全面资源,并强调了异质性髓系浸润的免疫抑制作用。这些发现也表明,将TIM-3阻断与MAPK抑制剂联合使用,可能成为同时靶向TME和致癌MAPK通路的潜在治疗策略。
12. A single-cell and spatial genomics atlas of human skin fibroblasts reveals shared disease-related fibroblast subtypes across tissues
人类皮肤成纤维细胞的单细胞与空间基因组学图谱揭示了跨组织共存的疾病相关成纤维细胞亚型

英国剑桥大学威康桑格研究所
Fibroblasts sculpt the architecture and cellular microenvironments of various tissues. Here we constructed a spatially resolved atlas of human skin fibroblasts from healthy skin and 23 skin diseases, with comparison to 14 cross-tissue diseases. We define six major skin fibroblast subtypes in health and three that are disease-specific. We characterize two fibroblast subtypes further as they are conserved across tissues and are immune-related. The first, F3: fibroblastic reticular cell-like fibroblast (CCL19+CD74+HLA-DRA+), is a fibroblastic reticular cell-like subtype that is predicted to maintain the superficial perivascular immune niche. The second, F6: inflammatory myofibroblasts (IL11+MMP1+CXCL8+IL7R+), characterizes early human skin wounds, inflammatory diseases with scarring risk and cancer. F6: inflammatory myofibroblasts were predicted to recruit neutrophils, monocytes and B cells across multiple human tissues. Our study provides a harmonized nomenclature for skin fibroblasts in health and disease, contextualized with cross-tissue findings and clinical skin disease profiles.
成纤维细胞塑造了多种组织的结构与细胞微环境。该研究构建了一个空间分辨率的人类皮肤成纤维细胞图谱,涵盖健康皮肤及23种皮肤疾病,并与14种跨组织疾病进行了比较分析。该研究定义了健康状态下的六大主要皮肤成纤维细胞亚型,以及三种疾病特异性亚型。研究人员进一步鉴定了两种在跨组织间保守且与免疫功能相关的成纤维细胞亚型:第一种是F3亚型(成纤维细胞性网状细胞样成纤维细胞,标志物为CCL19+CD74+HLA-DRA+),该亚型具有成纤维细胞性网状细胞特征,被预测为维持浅层血管周免疫微环境的关键细胞;第二种是F6亚型(炎性肌成纤维细胞,标志物为IL11+MMP1+CXCL8+IL7R+),其特征性存在于早期人类皮肤创面、具有瘢痕风险的炎症性疾病及癌症中。预测分析显示,F6炎性肌成纤维细胞在多种人类组织中具有招募中性粒细胞、单核细胞和B细胞的能力。该研究为健康与疾病状态下的皮肤成纤维细胞建立了统一命名体系,并结合跨组织研究结果与临床皮肤病特征进行了系统性阐释。
13. Divergent cytokine and transcriptional signatures control functional T follicular helper cell heterogeneity
差异化的细胞因子与转录特征调控滤泡辅助性T细胞的功能异质性

澳大利亚沃尔特与伊丽莎·霍尔医学研究所
CD4+ T follicular helper (TFH) cells support tailored B cell responses against multiple classes of pathogens. To reveal how diverse TFH phenotypes are established, we profiled mouse TFH cells in response to viral, helminth and bacterial infection. We identified a core TFH signature that is distinct from CD4+ T follicular regulatory and effector cells and identified pathogen-specific transcriptional modules that shape TFH function. Cytokine-transcriptional TFH programming demonstrated that type I interferon and TGFβ signaling direct individual TFH phenotypes to instruct B cell output. Cytokine-directed TFH transcriptional phenotypes are shared within human germinal centers, but distinct TFH phenotypes dominate between donors and following immune challenge or in antibody-mediated disease. Finally, we identified new cell surface markers that align with distinct TFH phenotypes. Thus, we provide a comprehensive resource of TFH diversity in humans and mice to enable immune monitoring during infection and disease and to inform the development of context-specific vaccines.
CD4⁺滤泡辅助性T细胞(TFH细胞)支持针对多种病原体的特异性B细胞应答。为揭示多样化的TFH细胞表型如何建立,研究人员对小鼠TFH细胞在病毒、蠕虫和细菌感染中的应答进行了系统分析。研究鉴定出一个区别于CD4⁺滤泡调节性T细胞和效应T细胞的核心TFH特征谱,并确定了塑造TFH细胞功能的病原体特异性转录模块。通过细胞因子-转录联合编程分析发现,I型干扰素和TGFβ信号通路可指导个体TFH细胞表型形成,进而调控B细胞的功能输出。这种细胞因子导向的TFH转录表型在人类生发中心中同样存在,但在不同供体间、免疫刺激后或抗体介导疾病中占主导的TFH表型存在显著差异。最后,该研究鉴定出与不同TFH表型相匹配的新型细胞表面标志物。该研究为人类和小鼠的TFH细胞多样性提供了全面的资源库,可用于感染与疾病期间的免疫监测,并为开发情境特异性疫苗提供理论依据。
Volume 26, Issue 11
本期共发表论著12篇。
1. Hypoxia induces histone clipping and H3K4me3 loss in neutrophil progenitors resulting in long-term impairment of neutrophil immunity
低氧通过诱导中性粒细胞前体发生组蛋白剪切及H3K4me3丢失,导致中性粒细胞免疫功能长期受损

爱丁堡大学再生与修复研究所、拉德堡德大学医学中心
The long-term impact of systemic hypoxia resulting from acute respiratory distress syndrome (ARDS) on the function of short-lived innate immune cells is unclear. We show that patients 3–6 months after recovering from ARDS have persistently impaired circulating neutrophil effector functions and an increased susceptibility to secondary infections. These defects are linked to a widespread loss of the activating histone mark H3K4me3 in genes that are crucial for neutrophil activities. By studying healthy volunteers exposed to altitude-induced hypoxemia, we demonstrate that oxygen deprivation alone causes this long-term neutrophil reprogramming. Mechanistically, mouse models of systemic hypoxia reveal that persistent loss of H3K4me3 originates in proNeu and preNeu progenitors within the bone marrow and is linked to N-terminal histone 3 clipping, which removes the lysine residue for methylation. Thus, we present new evidence that systemic hypoxia initiates a sustained maladaptive reprogramming of neutrophil immunity by triggering histone 3 clipping and H3K4me3 loss in neutrophil progenitors.
急性呼吸窘迫综合征(ARDS)引发的全身性低氧对短寿命固有免疫细胞功能的长期影响尚不明确。该研究发现,在ARDS康复后3–6个月的患者体内,循环中性粒细胞的效应功能仍持续受损,且继发感染易感性增高。这些缺陷与中性粒细胞功能关键基因中激活型组蛋白标记H3K4me3的广泛丢失相关。通过对暴露于高原低氧血症的健康志愿者进行研究,研究证实单纯氧剥夺即可导致这种长期的中性粒细胞重编程。机制研究表明,全身性低氧小鼠模型揭示H3K4me3的持续性丢失起源于骨髓中的proNeu和preNeu前体细胞,并与组蛋白H3 N端剪切现象相关——该剪切过程移除了可被甲基化的赖氨酸位点。因此,研究提出了新的证据:全身性低氧通过触发中性粒细胞前体中的组蛋白H3剪切及H3K4me3丢失,启动了中性粒细胞免疫功能的持续性适应不良性重编程。
2. STING signals to NF-κB from late endolysosomal compartments using IRF3 as an adaptor
STING通过IRF3作为衔接蛋白,在晚期内吞溶酶体区室中向NF-κB传导信号

丹麦奥胡斯大学生物医学系
Maturation of conventional dendritic cells (cDCs) is crucial for maintaining tolerogenic safeguards against auto-immunity and for promoting immunogenic responses to pathogens and cancer. The subcellular mechanism for cDC maturation remains poorly defined. We show that cDCs mature by leveraging an internal reservoir of cholesterol (harnessed from extracellular cell debris and generated by de novo synthesis) to assemble lipid nanodomains on cell surfaces of maturing cDCs, enhance expression of maturation markers and stabilize immune receptor signaling. This process is dependent on cholesterol transport through Niemann–Pick disease type C1 (NPC1) and mediates homeostatic and Toll-like receptor (TLR)-induced maturation. Importantly, we identified the receptor tyrosine kinase AXL as a regulator of the NPC1-dependent construction of lipid nanodomains. Deleting AXL from cDCs enhances their maturation, thus improving anti-tumor immunity. Altogether, our study presents new insights into cholesterol mobilization as a fundamental basis for cDC maturation and highlights AXL as a therapeutic target for modulating cDCs.
NF-κB是免疫应答激活的核心调控因子。胞质DNA通过激活cGAS–STING通路诱导I型干扰素(IFNs)产生并启动NF-κB信号传导,从而激发宿主防御反应与病理性炎症。然而,STING诱导NF-κB激活的具体机制尚不明确。该研究发现,STING在高尔基体转运至内吞溶酶体区室后,以延迟方式激活NF-κB。该激活过程依赖干扰素诱导转录因子IRF3,但不依赖于I型干扰素信号通路,且这一激活模式在四足动物中具有进化保守性。机制研究表明,单体IRF3以相对于其与STING pS366位点结合(促进I型干扰素应答)更迟缓的动力学特征,被招募至STING的pS358位点。IRF3与STING pS358的结合诱导其向晚期内吞溶酶体区室转运,进而支持TRAF6的募集和NF-κB的激活。研究人员在IRF3中鉴定出一个TRAF6结合基序,该基序促进TRAF6的招募。这项工作定义了STING上的信号传导表面以及IRF3作为免疫信号衔接蛋白的新功能。这些发现表明,STING向NF-κB的信号传导仅能在其离开高尔基体后至溶酶体降解前的短暂时间窗口内实现,这可能有助于在稳态和危险感知条件下限制炎症反应的过度激活。
3. Chemotherapy-induced CA-repeat DNA fragments in breast cancer trigger antitumor immune responses
化疗诱导的乳腺癌CA重复序列DNA片段触发抗肿瘤免疫应答

中山大学孙逸仙纪念医院恶性肿瘤表观遗传与基因调控重点实验室、乳腺肿瘤中心
Damage-associated molecular patterns generated by cancer treatment can modulate antitumor immunity, but the underlying mechanisms of this effect are unclear. Here we show that CA-enriched DNA fragments resulting from DNA-damaging chemotherapy in MSH2-low tumors preferentially bind cGAS with strong affinity and form biomolecular condensates by phase separation in the cytoplasm, resulting in antitumor immunity. However, classical CA-poor DNAs released from MSH2-high tumor cells engage AIM2, resulting in immunosuppression by upregulating PD-L1 and IDO. Intratumoral administration of CA-rich DNA fragments enhanced antitumor immunity in syngrafted PyMT tumors. Clinically, CA-rich DNA abundance in breast cancer following chemotherapy was associated with increased tumor-antigen-reactive T cells and better chemotherapeutic responses. Therefore, different tumor DNA fragments can trigger opposing immune responses depending on their preference for differential sensors. This study highlights another mechanistic link between genome instability and immune modulation and the therapeutic potential of CA-rich DNAs to enhance antitumor immunity.
癌症治疗产生的损伤相关分子模式可调控抗肿瘤免疫,但其作用机制尚不明确。该研究发现,在MSH2低表达的肿瘤中,DNA损伤性化疗产生的富含CA的DNA片段能以高亲和力优先结合cGAS,并通过胞质内的相分离形成生物分子凝聚体,从而引发抗肿瘤免疫。然而,从MSH2高表达肿瘤细胞释放的经典贫CA DNA则会激活AIM2,通过上调PD-L1和IDO导致免疫抑制。瘤内注射富含CA的DNA片段可增强同种移植PyMT肿瘤模型的抗肿瘤免疫。临床数据显示,乳腺癌患者化疗后组织中富含CA的DNA丰度与肿瘤抗原反应性T细胞的增加及更好的化疗反应相关。因此,不同的肿瘤DNA片段可根据其对不同传感器的偏好性触发相反的免疫应答。这项研究揭示了基因组不稳定性与免疫调控之间的又一机制联系,并凸显了富含CA的DNA在增强抗肿瘤免疫方面的治疗潜力。
4. Plasmacytoid dendritic cells are dispensable or detrimental in murine systemic or respiratory viral infections
浆细胞样树突状细胞在小鼠全身性或呼吸道病毒感染中是非必需甚至有害的

艾克斯-马赛大学,法国国家科学研究中心
Plasmacytoid dendritic cells (pDCs) are major producers of type I/III interferons. As interferons are crucial for antiviral defense, pDCs are assumed to play an essential role in this process; however, robust evidence supporting this dogma is scarce. Genetic or pharmacological manipulations that eliminate pDCs or disrupt their interferon production often affect other cells, confounding interpretation. Here, to overcome this issue, we engineered pDC-less mice that are specifically and constitutively devoid of pDCs by expressing diphtheria toxin under coordinated control of the Siglech and Pacsin1 genes, uniquely coexpressed in pDCs. pDC-less mice mounted protective immunity against systemic infection with mouse cytomegalovirus and showed higher survival and less lung immunopathology to intranasal infection with influenza virus and SARS-CoV-2. Thus, contrary to the prevailing dogma, we revealed that pDCs and their interferons are dispensable or deleterious during several viral infections. pDC-less mice will enable rigorously reassessing the roles of pDCs in health and disease.
浆细胞样树突状细胞(pDCs)是I/III型干扰素的主要产生细胞。由于干扰素对抗病毒防御至关重要,pDCs通常被认为在此过程中发挥关键作用;然而,支持这一传统观点的确凿证据却很少。消除pDCs或破坏其干扰素产生的遗传或药物操作常会同时影响其他细胞,导致结论混淆。为解决这一问题,通过构建在pDCs特异性共表达的Siglech和Pacsin1基因协调调控下表达白喉毒素的小鼠,成功制备了特异性且持续缺失pDCs的小鼠模型。pDC缺失小鼠在对小鼠巨细胞病毒全身性感染中能建立保护性免疫,且在甲型流感病毒和SARS-CoV-2鼻内感染模型中表现出更高的生存率和更轻的肺部免疫病理损伤。因此,与主流观点相反,研究发现pDCs及其产生的干扰素在多种病毒感染过程中是非必要甚至有害的。pDC缺失小鼠模型将有助于重新严谨评估pDCs在健康与疾病中的作用。
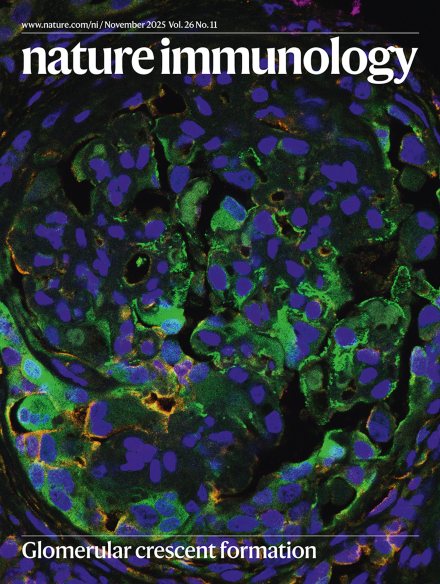
5. Spatiotemporal interaction of immune and renal cells controls glomerular crescent formation in autoimmune kidney disease
免疫细胞与肾脏细胞的时空相互作用调控自身免疫性肾脏疾病中的肾小球新月体形成

德国汉堡-埃彭多夫大学医学中心
Rapidly progressive glomerulonephritis (RPGN) is the most aggressive group of autoimmune kidney diseases and is characterized by glomerular crescent formation with proliferation of parietal epithelial cells (PECs). However, the underlying mechanisms of glomerular crescent formation are incompletely understood. Here we provide a high-resolution spatial kidney cell atlas of 57 samples from patients with RPGN (ANCA-associated GN, lupus nephritis and anti-glomerular basement membrane-GN) to characterize the cell signaling pathways in glomerular crescent development. Early platelet-derived growth factor (PDGF) signaling from epithelial and mesangial cells caused PEC activation and proliferation in glomerular crescents, whereas later transforming growth factor (TGF)-β signaling from macrophages, T cells and epithelial and mesangial cells triggered expression of extracellular matrix components in PECs associated with glomerulosclerosis and disease progression. These findings were similar across the different GNs and were functionally validated in experimental GN by PDGF and TGFβ blockade. These results highlight a spatiotemporally conserved progression program into glomerular crescents and sclerosis and indicate new treatment options for autoimmune kidney disease.
快速进展性肾小球肾炎(RPGN)是最具侵袭性的自身免疫性肾脏疾病,其特征为肾小球新月体形成伴随壁层上皮细胞(PECs)增殖。然而,肾小球新月体形成的潜在机制尚未完全阐明。该研究通过对57例RPGN患者(包括ANCA相关性肾炎、狼疮性肾炎和抗肾小球基底膜肾炎)样本进行高分辨率空间肾脏细胞图谱分析,系统揭示了肾小球新月体发育过程中的细胞信号通路。研究发现,早期由上皮细胞和系膜细胞产生的血小板衍生生长因子(PDGF)信号通路驱动了肾小球新月体中PECs的活化与增殖;而后期源自巨噬细胞、T细胞以及上皮细胞和系膜细胞的转化生长因子(TGF)-β信号通路则触发PECs表达细胞外基质成分,这一过程与肾小球硬化和疾病进展密切相关。这些发现在不同类型的肾小球肾炎中表现相似,并通过PDGF和TGFβ阻断实验在实验性肾炎模型中得到功能验证。该研究揭示了肾小球新月体与硬化形成的时空保守性进展程序,并为自身免疫性肾脏疾病指出了新的治疗方向。
6. Transcriptional and epigenetic targets of MEF2C in human microglia contribute to cellular functions related to autism risk and age-related disease
MEF2C在人类小胶质细胞中的转录与表观遗传学靶点参与调控自闭症风险及年龄相关疾病的细胞功能

加州大学圣迭戈分校儿科学
MEF2C encodes a transcription factor that is critical in nervous system development. Here, to examine disease-associated functions of MEF2C in human microglia, we profiled microglia differentiated from isogenic MEF2C-haploinsufficient and MEF2C-knockout induced pluripotent stem cell lines. Complementary transcriptomic and functional analyses revealed that loss of MEF2C led to a hyperinflammatory phenotype with broad phagocytic impairment, lipid accumulation, lysosomal dysfunction and elevated basal inflammatory cytokine secretion. Genome-wide profiling of MEF2C-bound sites coupled with the active regulatory landscape enabled inference of its transcriptional functions and potential mechanisms for MEF2C-associated cellular functions. Transcriptomic and epigenetic approaches identified substantial overlap with idiopathic autism datasets, suggesting a broader role of human microglial MEF2C dysregulation in idiopathic autism. In a mouse xenotransplantation model, loss of MEF2C led to morphological, lysosomal and lipid abnormalities in human microglia in vivo. Together, these studies reveal mechanisms by which reduced microglial MEF2C could contribute to the development of neurological diseases.
MEF2C基因编码一种对神经系统发育至关重要的转录因子。为探究MEF2C在人类小胶质细胞中的疾病相关功能,该研究对从同基因型MEF2C单倍体不足及MEF2C敲除诱导多能干细胞系分化的小胶质细胞进行了系统分析。互补的转录组学与功能分析显示,MEF2C缺失导致小胶质细胞呈现高炎症表型,表现为广泛的吞噬功能受损、脂质蓄积、溶酶体功能障碍以及基础炎症性细胞因子分泌升高。通过全基因组范围的MEF2C结合位点图谱分析,并结合活跃的染色质调控景观,研究人员推断出MEF2C的转录功能及其介导细胞功能的潜在机制。转录组学与表观遗传学方法揭示了其与特发性自闭症数据集存在显著重叠,提示人类小胶质细胞MEF2C失调在特发性自闭症中可能发挥更广泛的作用。在小鼠异种移植模型中,MEF2C缺失导致体内人类小胶质细胞出现形态异常、溶酶体及脂质代谢紊乱。综上,这些研究揭示了小胶质细胞MEF2C功能降低参与神经系统疾病发展的潜在机制。
7. Transient tissue residency and lymphatic egress define human CD56bright NK cell homeostasis
瞬时性组织驻留与淋巴管外排共同决定人类CD56bright NK细胞的稳态维持

卡罗林斯卡大学医院感染医学中心
Human tissue-resident (TR) CD56bright natural killer (NK) cells can be identified by expression of integrins and chemokine receptors inferred from murine studies, but many aspects of their homeostasis are unclear. Here we used an integrated approach of dynamic human, humanized mouse and non-human primate models and sampling of efferent lymph fluid to determine recirculation and TR patterns of human NK cells. By intravascular labeling, we showed that CD56bright NK cells access tissue niches at steady state. Furthermore, in human liver transplantation, donor-derived CD56bright NK cells represent the dominant TR NK cell population early after transplantation, but are replaced over time by infiltrating recipient NK cells that establish TR traits, a process partly regulated by Runx3. Transient TR CD56bright NK cells recirculated via lymphatics, displaying a consistent phenotype detectable in draining lymph nodes and efferent lymph fluid, and waned from peripheral blood on lymph node egress blockade. Finally, CD56dim NK cells, constrained to vasculature at steady state, entered lymph nodes upon inflammation. This study provides a mechanistic framework for the transient tissue residency and recirculation pattern of human NK cell populations.
人体组织驻留性(TR)CD56bright自然杀伤细胞(NK细胞)可通过整合素和趋化因子受体的表达来识别——这些标志物是从小鼠研究中推演而来,但其稳态维持的诸多方面仍不清楚。该研究通过整合动态人体模型、人源化小鼠模型及非人灵长类模型,并结合输出淋巴液采样,系统阐明了人类NK细胞的再循环与组织驻留模式。通过血管内标记技术,研究发现CD56bright NK细胞在稳态下可进入组织微环境。进一步在人类肝移植模型中观察到,供体来源的CD56bright NK细胞在移植早期占主导地位,但随时间推移逐渐被具有组织驻留特征的受者NK细胞所替代——该过程部分受Runx3调控。瞬时性组织驻留CD56bright NK细胞可通过淋巴管再循环,在引流淋巴结和输出淋巴液中检测到其稳定表型,且在淋巴结流出受阻时从外周血中减少。最后,通常局限于血管系统的CD56dim NK细胞在炎症状态下可进入淋巴结。这项研究为人类NK细胞群体的瞬时组织驻留与再循环模式提供了机制性框架。
8. Profiling of HIV-1 elite neutralizer cohort reveals a CD4bs bnAb for HIV-1 prevention and therapy
通过对HIV-1精英中和者队列的系统分析,鉴定出可用于HIV-1预防与治疗的CD4结合位点广谱中和抗体

科隆大学实验免疫学实验室
Administration of HIV-1 neutralizing antibodies can suppress viremia and prevent infection in vivo. However, clinical use is challenged by envelope diversity and rapid viral escape. Here, we performed single B cell profiling of 32 top HIV-1 elite neutralizers to identify broadly neutralizing antibodies with highest antiviral activity. From 831 expressed monoclonal antibodies, we identified 04_A06, a VH1-2-encoded broadly neutralizing antibody to the CD4 binding site with remarkable breadth and potency against multiclade pseudovirus panels (geometric mean half-maximal inhibitory concentration = 0.059 µg ml−1, breadth = 98.5%, 332 strains). Moreover, 04_A06 was not susceptible to classic CD4 binding site escape variants and maintained full viral suppression in HIV-1-infected humanized mice. Structural analyses revealed an unusually long 11-amino-acid heavy chain insertion that facilitates interprotomer contacts with highly conserved residues on the adjacent gp120 protomer. Finally, 04_A06 demonstrated high activity against contemporaneously circulating viruses from the Antibody-Mediated Prevention trials (geometric mean half-maximal inhibitory concentration = 0.082 µg ml−1, breadth = 98.4%, 191 virus strains), and in silico modeling for 04_A06LS predicted prevention efficacy of >93%. Thus, 04_A06 will provide unique opportunities for effective treatment and prevention of HIV-1 infection.
给予HIV-1中和抗体可抑制病毒血症并预防体内感染,但其临床应用受到病毒包膜多样性和快速免疫逃逸的挑战。该研究通过对32名顶尖HIV-1精英中和者进行单B细胞分析,旨在鉴定具有最强抗病毒活性的广谱中和抗体。从831个表达的单克隆抗体中,研究鉴定出04_A06——一种由VH1-2基因编码的靶向CD4结合位点的广谱中和抗体,其对多亚型假病毒组合表现出卓越的广谱性与中和效力(几何平均半抑制浓度=0.059µg·ml⁻¹,广谱性=98.5%,覆盖332种毒株)。此外,04_A06对经典的CD4结合位点逃逸变异株不敏感,并在HIV-1感染的人源化小鼠模型中维持完全病毒抑制。结构分析揭示其重链存在一个异常长的11个氨基酸插入,该插入促进了与相邻gp120原聚体上高度保守残基的原聚体间接触。最后,04_A06对来自抗体介导预防试验的当代流行毒株显示出高效力(几何平均半抑制浓度=0.082µg·ml⁻¹,广谱性=98.4%,覆盖191种病毒),计算机模拟预测04_A06LS变体的预防效能>93%。因此,04_A06将为HIV-1感染的有效治疗与预防提供独特机遇。
9. Spatial and functional diversity of innate lymphoid cells in the human female genital tract may contribute to antiviral responses to HIV
人类女性生殖道中先天淋巴细胞的空间与功能多样性可能有助于针对HIV的抗病毒应答

波士顿塔夫茨大学医学院免疫学、韦恩州立大学医学院微生物学与免疫学系
Innate lymphoid cells (ILCs) are tissue-resident lymphocytes specialized in cytokine secretion that lack antigen-specific receptors. The contribution of ILCs to antiviral mucosal immunity in humans, particularly in the female genital tract (FGT), remains unexplored. Here we resolved human FGT ILC diversity by spectral flow cytometry and CITE-seq, spatial location within genital anatomical regions using ChipCytometry, and determined homeostatic function and antiviral responses. We uncovered spatial and functional specializations of genital ILC subsets under homeostasis, with compartmentalized age-related and pregnancy-related changes. CD161 expression differentially discriminated ILC subsets preloaded with cytokines at steady state. We identified a unique NKp44+CCR6+ ILC3 subset in the endometrium that actively degranulated at homeostasis and was located in lymphoid aggregates surrounded by B cells and T cells. By contrast, ILC1s were found scattered, enriched in the ectocervix and located close to the epithelium. Following in vitro HIV stimulation, genital ILCs displayed rapid subset-specific antiviral responses. These findings reveal distinct tissue and subset-specific features of FGT ILCs and their capacity to immediately respond to viral stimuli, providing a foundation for future studies to determine the potential role of ILCs in mucosal immune protection in the FGT.
先天淋巴细胞(ILCs)是一类缺乏抗原特异性受体的组织驻留性淋巴细胞,其功能特化为细胞因子分泌。ILCs在人类抗病毒黏膜免疫——特别是女性生殖道(FGT)免疫中的作用尚未被探索。该研究通过光谱流式细胞术与CITE-seq技术解析了人类FGT中ILC的多样性,利用ChipCytometry技术明确了其在生殖解剖区域的空间定位,并确定了其稳态功能与抗病毒应答。研究揭示了稳态条件下生殖道ILC亚群的空间与功能特化特征,以及随年龄与妊娠状态发生的区室化变化。CD161表达差异性地识别了在稳态下预载细胞因子的ILC亚群。研究人员在子宫内膜中发现了一个独特的NKp44⁺CCR6⁺ ILC3亚群,该亚群在稳态下即处于活跃的脱颗粒状态,并定位于由B细胞和T细胞包围的淋巴聚集区。相比之下,ILC1s呈散在分布,在外宫颈富集且定位于上皮附近。在体外HIV刺激后,生殖道ILCs显示出快速的亚群特异性抗病毒应答。这些发现揭示了FGT中ILC组织与亚群特异性特征及其对病毒刺激的即时响应能力,为未来研究ILCs在FGT黏膜免疫保护中的潜在作用奠定了基础。
10. Influenza vaccine based on AS03-adjuvanted chimeric HA induces long-lived stalk-specific plasma cells in bone marrow and lymph nodes of nonhuman primates
基于AS03佐剂嵌合HA的流感疫苗可在非人灵长类动物骨髓与淋巴结中诱导长寿命茎部特异性浆细胞生成

埃默里大学埃默里国家灵长类动物研究中心、微生物学与免疫学
Current influenza vaccines face challenges due to antigenic evolution of the circulating virus and waning immunity in humans. Here we investigated the durability of humoral immunity induced by an influenza vaccine based on AS03-adjuvanted chimeric hemagglutinin (cHA) in nonhuman primates (NHPs). Two groups of NHPs received two doses of a seasonal quadrivalent influenza vaccine, followed by sequential immunization with split virus cHA vaccines cH8/1N1, and cH5/1N1. One group received cHA immunizations with AS03 adjuvant. We monitored serum antibodies and long-lived plasma cells in bone marrow for nearly 2 years after the final vaccination. cHA vaccines induced stalk-specific antibody responses. The addition of AS03 enhanced both the magnitude and durability of humoral immunity by establishing long-lived plasma cells in the bone marrow and lymph nodes for nearly 2 years. Passive transfer of NHP serum provided protection against challenge with heterologous influenza A virus strains in mice. This study highlights the potential of the AS03-adjuvanted chimeric HA vaccine strategy to provide durable and broadly protective humoral immunity.
当前流感疫苗因病毒抗原性进化及人体免疫力衰减而面临挑战。该研究在非人灵长类动物(NHPs)中探究了基于AS03佐剂嵌合血凝素(cHA)的流感疫苗所诱导的体液免疫持久性。两组NHPs先接种两剂季节性四价流感疫苗,随后依次接种裂解病毒cHA疫苗cH8/1N1与cH5/1N1。其中一组接种时添加AS03佐剂。研究人员在最终疫苗接种后近两年内持续监测了血清抗体水平及骨髓中的长寿命浆细胞。cHA疫苗成功诱导了茎部特异性抗体应答。添加AS03佐剂通过促使长寿命浆细胞在骨髓和淋巴结中持续存在近两年,显著增强了体液免疫的强度与持久性。将NHP血清被动转移至小鼠后,能提供针对异源甲型流感病毒毒株攻击的保护作用。这项研究凸显了AS03佐剂嵌合HA疫苗策略在提供持久且广谱保护性体液免疫方面的潜力。
11. Temporal and context-dependent requirements for the transcription factor Foxp3 expression in regulatory T cells
转录因子Foxp3在调节性T细胞中的表达需求具有时间与情境依赖性

斯隆·凯特琳研究所、耶鲁大学免疫生物学系及生物分子设计与发现研究所
Regulatory T (Treg) cells, expressing the transcription factor Foxp3, are obligatory gatekeepers of immune responsiveness, yet the mechanisms by which Foxp3 governs the Treg transcriptional network remain incompletely understood. Using a novel chemogenetic system of inducible Foxp3 protein degradation in vivo, we found that while Foxp3 was indispensable for the establishment of transcriptional and functional programs of newly generated Treg cells, Foxp3 loss in mature Treg cells resulted in minimal functional and transcriptional changes under steady state. This resilience of the Foxp3-dependent program in mature Treg cells was acquired over an unexpectedly long timescale; however, in settings of severe inflammation, Foxp3 loss led to a pronounced perturbation of Treg cell transcriptome and fitness. Furthermore, tumoral Treg cells were uniquely sensitive to Foxp3 degradation, which led to impairment in their suppressive function and tumor shrinkage in the absence of pronounced adverse effects. These studies demonstrate a context-dependent differential requirement for Foxp3 for Treg transcriptional and functional programs.
表达转录因子Foxp3的调节性T(Treg)细胞是免疫应答的必需调控者,但Foxp3如何支配Treg细胞转录网络的机制尚未完全阐明。通过使用一种新型的化学遗传学系统在体内诱导Foxp3蛋白降解,研究发现:虽然Foxp3对于新生成Treg细胞的转录与功能程序的建立不可或缺,但在稳态条件下,成熟Treg细胞中Foxp3的缺失仅导致极微小的功能与转录变化。这种Foxp3依赖程序的稳健性是在一个出乎意料的长时间尺度上逐渐获得的;然而,在严重炎症环境中,Foxp3的缺失会导致Treg细胞转录组及其适应性的显著紊乱。此外,肿瘤内Treg细胞对Foxp3降解表现出独特的敏感性,这导致其抑制功能受损并在未引起明显副作用的情况下促进肿瘤萎缩。这些研究揭示了Foxp3对Treg细胞转录与功能程序的需求具有环境依赖性差异。
12. The co-inhibitory receptor TIGIT promotes tissue-protective functions in T cells
共抑制性受体TIGIT可促进T细胞发挥组织保护功能

苏黎世大学定量生物医学系
The co-inhibitory receptor TIGIT suppresses excessive immune responses in autoimmune conditions while also restraining antitumor immunity. In viral infections, TIGIT alone does not affect viral control but has been shown to limit tissue pathology. However, the underlying mechanisms are incompletely understood. Here we found TIGIT+ T cells to express not only an immunoregulatory gene signature but also a tissue repair gene signature. Specifically, after viral infection, TIGIT directly drives expression of the tissue growth factor amphiregulin (Areg), which is strongly reduced in the absence of TIGIT. We identified regulatory T (Treg) cells, but not CD8+ T cells, as the critical T cell subset mediating these tissue-protective effects. In Treg cells, TIGIT engagement after T cell antigen receptor stimulation induces the transcription factor Blimp-1, which then promotes Areg production and tissue repair. Thus, we uncovered a nonclassical function of the co-inhibitory receptor TIGIT, wherein it not only limits immune pathology by suppressing the immune response but also actively fosters tissue regeneration by inducing the tissue growth factor Areg in T cells.
共抑制性受体TIGIT在自身免疫性疾病中抑制过度的免疫应答,同时也限制抗肿瘤免疫。在病毒感染中,仅TIGIT本身不影响病毒控制,但已被证明能限制组织病理损伤,然而其潜在机制尚未完全阐明。该研究发现,TIGIT⁺ T细胞不仅表达免疫调节基因特征,还表达组织修复基因特征。具体而言,病毒感染后,TIGIT直接驱动组织生长因子双调蛋白(Areg)的表达,该因子在TIGIT缺失时显著减少。研究确定调节性T(Treg)细胞——而非CD8⁺ T细胞——是介导这些组织保护作用的关键T细胞亚群。在Treg细胞中,T细胞抗原受体刺激后TIGIT的激活可诱导转录因子Blimp-1,进而促进Areg产生和组织修复。因此,研究揭示了共抑制性受体TIGIT的一种非经典功能:它不仅通过抑制免疫应答来限制免疫病理损伤,还通过诱导T细胞中的组织生长因子Areg来主动促进组织再生。
Volume 26, Issue 12
本期共发表论著12篇。
1. Sequential lymphotoxin-β receptor and retinoic acid receptor signals regulate cDC2 fate
淋巴毒素-β受体与视黄酸受体的级联信号传导共同调控cDC2细胞命运决定

多伦多大学免疫学系、霍华德·休斯医学研究所
Type 2 conventional dendritic cells (cDC2s) are functionally and phenotypically heterogenous. Previous work in mice and humans identified two cDC2 subsets (cDC2As and cDC2Bs) and a monocytic DC3 subset. However, the microenvironmental cues governing their distinct differentiation pathways remain unclear. Here, we delineate mouse cDC2 lineage relationships and the sequential signals required for cDC2A maintenance. We show that cDC2s, arising from the CLEC9A+ cDC progenitor, encompass T-BET-expressing cDC2As and two cDC2B subsets distinguished by MGL2 expression, with monocytic DC3s exhibiting transcriptional overlap with Mgl2− cDC2Bs. Among these subsets, T-BET+ cDC2As dominate the spleen where they require cell-intrinsic retinoic acid signaling to sustain their differentiation via Notch signals. Lymphotoxin-β receptor signaling on splenic cDC2s limits F-actin content retaining cDC2s at sites of retinol delivery. In summary, these data establish the developmental and transcriptional relationships between diverse cDC2 subsets and identify signals that regulate their prevalence in specific lymphoid tissues.
2型传统树突状细胞(cDC2s)在功能和表型上具有异质性。先前在小鼠和人类中的研究已鉴定出两个cDC2亚群(cDC2A和cDC2B)以及一个单核细胞来源的DC3亚群。然而,调控其不同分化途径的微环境信号仍不明确。该研究系统阐明了小鼠cDC2的谱系关系以及维持cDC2A所需的级联信号。研究发现,来源于CLEC9A⁺ cDC前体细胞的cDC2s包含表达T-BET的cDC2A和两个以MGL2表达区分的cDC2B亚群,而单核细胞来源的DC3s在转录谱上与Mgl2⁻ cDC2B存在重叠。在这些亚群中,T-BET⁺ cDC2A在脾脏中占主导地位,其需要通过细胞固有的视黄酸信号传导,经由Notch信号通路维持分化状态。脾脏cDC2s上的淋巴毒素-β受体信号可通过限制F-肌动蛋白含量,将cDC2s滞留于视黄醇递送部位。总之,这些数据确立了不同cDC2亚群之间的发育与转录关系,并鉴定了调控其在特定淋巴组织中丰度的关键信号。
2. Fibroblastic FLT3L supports lymph node dendritic cells in the interfollicular niche
成纤维细胞来源的FLT3L通过滤泡间微环境维持淋巴结树突状细胞稳态

美国Genentech眼科与免疫学部、蛋白质组学与基因组技术部
Dendritic cell (DC) homeostasis is maintained in secondary lymphoid organs (SLOs) by Fms-like tyrosine kinase 3 ligand (FLT3L). The specific niche providing this DC growth factor within human and mouse SLOs is unclear. Here we show that Gremlin1 (GREM1)-expressing lymph node fibroblastic reticular cells (FRCs) support DC homeostasis via provision of FLT3L. Grem1-expressing FRCs colocalize with DCs and express FLT3LG/Flt3l in human and mouse lymph nodes. Using a new genetic model, we provide evidence that FLT3L produced by GREM1+ FRCs maintains lymph node DC precursors (preDCs) and both conventional (cDCs) and plasmacytoid DCs (pDCs). Spatial transcriptomics and cytofluorometry reveal that GREM1+ FRC-derived FLT3L supports not only proliferation, but also survival of lymph node preDCs and cDCs within the interfollicular zone (IFZ). Functionally, loss of GREM1+ FRC-derived FLT3L impairs cDC activation of antigen-specific T cell responses to both immunization and infection. These findings provide key mechanistic insights underlying stromal cell support of DC homeostasis and function.
树突状细胞(DCs)在次级淋巴器官(SLOs)中的稳态由Fms样酪氨酸激酶3配体(FLT3L)维持。然而,在人类和小鼠SLOs中提供该DC生长因子的特定微环境尚不明确。该研究发现,表达Gremlin1(GREM1)的淋巴结成纤维细胞性网状细胞(FRCs)通过提供FLT3L来支持DC稳态。Grem1表达的FRCs在人类和小鼠淋巴结中与DCs共定位并表达FLT3LG/Flt3l基因。利用新型遗传模型,研究证实GREM1⁺ FRCs产生的FLT3L可维持淋巴结DC前体细胞(preDCs)以及传统DCs(cDCs)和浆细胞样DCs(pDCs)的稳态。空间转录组学与细胞流式分析揭示,GREM1⁺ FRC来源的FLT3L不仅支持滤泡间区(IFZ)内淋巴结preDCs和cDCs的增殖,还维持其存活。在功能上,缺失GREM1⁺ FRC来源的FLT3L会损害cDCs在免疫接种和感染过程中对抗原特异性T细胞应答的激活能力。这些发现为基质细胞支持DC稳态与功能的机制提供了关键见解。
3. Cancer suppresses mitochondrial chaperone activity in macrophages to drive immune evasion
肿瘤通过抑制巨噬细胞中的线粒体分子伴侣活性驱动免疫逃逸

俄亥俄州立大学医学院、洛桑大学
Contrary to tumor-infiltrating T cells with dysfunctional mitochondria, tumor-associated macrophages (TAMs) preserve their mitochondrial activity in the nutrient-limited tumor microenvironment (TME) to sustain immunosuppression. Here we identify TNF receptor-associated protein-1 (TRAP1), a mitochondrial HSP90 chaperone, as a metabolic checkpoint that restrains oxidative respiration and limits macrophage suppressive function. In the TME, TRAP1 is downregulated through TIM4–AMPK signaling, and its loss enhances immunoinhibitory activity, limits proinflammatory capacity and promotes tumor immune escape. Mechanistically, TRAP1 suppression augments electron transport chain activity and elevates the α-ketoglutarate/succinate ratio, remodeling mitochondrial homeostasis. The resulting accumulation of α-ketoglutarate further potentiates JMJD3-mediated histone demethylation, establishing transcriptional programs that reinforce an immunosuppressive state. Restoring TRAP1 by targeting TIM4 and JMJD3 reprograms TAMs, disrupts the immune-evasive TME and bolsters antitumor immunity. These findings establish TRAP1 as a critical regulator integrating metabolic and epigenetic control of suppressive TAM function and position the TRAP1 pathway as a promising target for cancer immunotherapy.
与线粒体功能障碍的肿瘤浸润T细胞不同,肿瘤相关巨噬细胞(TAMs)在营养受限的肿瘤微环境(TME)中仍能维持其线粒体活性以支持免疫抑制功能。该研究发现,线粒体HSP90分子伴侣TNF受体相关蛋白-1(TRAP1)是限制氧化呼吸并抑制巨噬细胞免疫抑制功能的代谢检查点。在TME中,TRAP1通过TIM4–AMPK信号通路被下调,其缺失会增强免疫抑制活性、限制促炎能力并促进肿瘤免疫逃逸。机制上,TRAP1抑制可增强电子传递链活性并提高α-酮戊二酸/琥珀酸比值,从而重塑线粒体内稳态。由此积累的α-酮戊二酸进一步强化JMJD3介导的组蛋白去甲基化,建立强化免疫抑制状态的转录程序。通过靶向TIM4和JMJD3恢复TRAP1表达可重编程TAMs,破坏免疫逃逸型TME并增强抗肿瘤免疫。这些发现确立了TRAP1作为整合抑制性TAM功能代谢与表观遗传调控的关键调节因子,并将TRAP1通路定位为癌症免疫治疗的重要靶点。
4. Interferon-mediated NK cell activation increases cytolytic activity against T follicular helper cells and limits antibody response to SARS-CoV-2
干扰素介导的NK细胞活化增强其对滤泡辅助性T细胞的杀伤活性,从而限制针对SARS-CoV-2的抗体应答

斯坦福大学医学院
Natural killer (NK) cells are innate lymphocytes known for their ability to kill infected or malignant cells, but they have an overlooked role in regulating antibody responses. In mice, NK cells can kill T follicular helper (TFH) cells, decreasing somatic hypermutation and antibody titers. Although human NK cell activation correlates with poor vaccine response, the mechanisms of NK cell regulation of adaptive immunity in humans are poorly understood. Here we found that, in ancestral severe acute respiratory syndrome coronavirus 2 infection, individuals with the broadest neutralization profile had fewer NK cells that expressed inhibitory and immaturity markers, whereas NK cells from narrow neutralizers were highly activated and expressed interferon-stimulated genes. ISG-mediated activation in NK cells from healthy donors increased cytotoxicity toward induced TFH-like cells via NKG2D and NKp30. This work reveals that NK cell activation and dysregulated inflammation play a role in poor antibody response to severe acute respiratory syndrome coronavirus 2 and opens exciting avenues for designing improved vaccines and adjuvants to target emerging pathogens.
自然杀伤细胞(NK细胞)是以杀伤感染或恶性细胞能力著称的先天淋巴细胞,但其在调节抗体应答中的作用常被忽视。在小鼠中,NK细胞可杀伤滤泡辅助性T细胞(TFH细胞),从而减少体细胞高频突变和抗体滴度。尽管人类NK细胞活化与疫苗应答不良相关,但其调控适应性免疫的机制尚不清楚。该研究发现,在原始型严重急性呼吸综合征冠状病毒2(SARS-CoV-2)感染中,具有最广谱中和能力的个体其NK细胞表达抑制性和不成熟标记的比例更低,而中和能力狭窄个体的NK细胞则高度活化并表达干扰素刺激基因(ISGs)。来自健康供体的NK细胞经ISG介导活化后,通过NKG2D和NKp30受体增强了对诱导型TFH样细胞的细胞毒性。这项工作揭示了NK细胞活化与失调性炎症在针对SARS-CoV-2的抗体应答不良中发挥作用,并为设计靶向新发病原体的改良疫苗和佐剂开辟了新方向。
5. TSLP links intestinal nutrient sensing with amplification of the ILC2–tuft cell circuit
胸腺基质淋巴细胞生成素(TSLP)将肠道营养感知与2型固有淋巴细胞-簇细胞回路的扩增相偶联

加州大学旧金山分校医学系、多伦多西奈山医院
Group 2 innate lymphocytes (ILC2s) are prevalent in small intestine but their role during homeostasis is unclear. Here we show that thymic stromal lymphopoietin (TSLP)—a cytokine implicated in ILC2 activation—is expressed constitutively in subepithelial fibroblasts, including telocytes and crypt-associated trophocytes, which are specialized fibroblasts necessary to sustain epithelial identity. Feeding increases TSLP and induces ILC2 type 2 cytokines that are attenuated by deletion of TSLP in fibroblasts or TSLP receptor on ILC2s. Both mouse and human intestinal fibroblasts express receptors for glucagon-like peptide-2 (GLP-2)—an intestinotrophic growth factor released by enteroendocrine cells following food intake. GLP-2 promotes intestinal TSLP in mouse and human intestinal fibroblasts, and TSLP-dependent ILC2 activation and tuft cell hyperplasia in mice, thus linking nutrient detection with ILC2-mediated amplification of the tuft cell chemosensory circuit that promotes epithelial surveillance of ingested cargo.
2型固有淋巴细胞(ILC2s)在小肠中大量存在,但其在稳态维持中的作用尚不明确。该研究发现,胸腺基质淋巴细胞生成素(TSLP)——一种参与ILC2活化的细胞因子——在上皮下层成纤维细胞(包括端细胞和隐窝相关滋养细胞)中组成型表达,这些特化成纤维细胞是维持上皮细胞特性所必需的。进食可增加TSLP表达并诱导ILC2产生2型细胞因子,该过程在成纤维细胞缺失TSLP或ILC2s缺失TSLP受体时被削弱。小鼠和人类肠道成纤维细胞均表达胰高血糖素样肽-2(GLP-2)受体——GLP-2是肠内分泌细胞在进食后释放的促肠道生长因子。GLP-2可促进小鼠和人类肠道成纤维细胞产生TSLP,并在小鼠中引发TSLP依赖性的ILC2活化与簇细胞增生,从而将营养感知与ILC2介导的簇细胞化学感应回路扩增相连接,该回路促进上皮对摄入物质的持续监视。
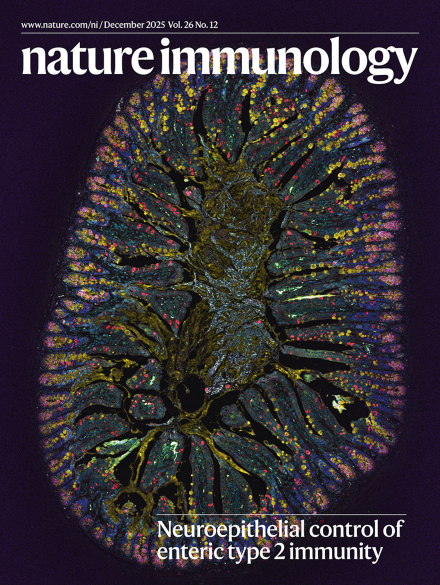
6. Enteric nervous system-derived VIP restrains differentiation of LGR5+ stem cells toward the secretory lineage impeding type 2 immune programs
肠道神经系统来源的血管活性肠肽通过限制LGR5⁺干细胞向分泌谱系分化来抑制2型免疫应答程序

柏林夏里特医学院、伯尔尼大学医院因塞尔医院
Barrier homeostasis relies on a finely tuned interplay between the immune system, epithelial cells and commensal microbiota. Beyond these regulators, the enteric nervous system has recently emerged as a central hub coordinating intestinal immune responses, although its role in epithelial differentiation has remained largely unexplored. Here, we identify a neuroepithelial circuit in which vasoactive intestinal peptide (VIP)-positive enteric neurons act on VIPR1+ epithelial stem cells to restrain both their proliferation and secretory lineage differentiation. Disruption of this pathway leads to an expansion of tuft cells, enhanced interleukin (IL)-25 production, activation of group 2 innate lymphoid cells (ILC2s) and induction of a type 2 immune response resembling worm expulsion. This phenotype occurs independently of the microbiota but is modulated by the IL-25R–ILC2–IL-13 axis and dietary solid food intake. Our findings expose the enteric nervous system as a critical regulator of epithelial fate decisions and immune balance, complementing established mechanisms that safeguard barrier integrity and mucosal homeostasis.
屏障稳态依赖于免疫系统、上皮细胞和共生微生物群之间精细调控的相互作用。除了这些调控因子,肠道神经系统最近被证实是协调肠道免疫应答的中心枢纽,但其在上皮分化中的作用仍很大程度上未被探索。该研究发现了一条神经上皮环路:血管活性肠肽(VIP)阳性的肠道神经元作用于VIPR1⁺上皮干细胞,抑制其增殖及向分泌谱系的分化。破坏该通路会导致簇细胞扩增、白细胞介素(IL)-25产生增加、2型固有淋巴细胞(ILC2s)活化,并诱导类似蠕虫驱除反应的2型免疫应答。这一表型的发生不依赖于微生物群,但受IL-25R–ILC2–IL-13轴及饮食中固体食物摄入的调节。研究揭示了肠道神经系统作为上皮细胞命运决定与免疫平衡的关键调节者,完善了现有维持屏障完整性与黏膜稳态的调控机制。
7. Neuroepithelial VIP–VIPR1 interactions differentially control enteric type 1 and type 2 immunity
神经上皮细胞间的VIP–VIPR1相互作用差异性地调控肠道1型与2型免疫应答

科英布拉大学、柏林夏里特医学院
The nervous and immune systems cooperate to regulate mucosal barrier integrity. Nevertheless, whether enteric neurons establish neuroepithelial interactions to coordinate immunity remains elusive. Here, we identified neuroepithelial interactions that differentially control intestinal type 1 and type 2 immunity. Gut epithelial cells expressed vasoactive intestinal peptide (VIP) receptor 1 (VIPR1), and chemogenetic modulation of enteric VIPergic neurons led to altered epithelial-derived cytokines. Epithelial-intrinsic deletion of Vipr1 resulted in diminished type 1 immunity, including reduced type 1 alarmins and intraepithelial lymphocytes. In contrast, epithelial Vipr1 deficiency led to enhanced type 2 immunity, comprising increased type 2 alarmins, tuft cells and activated group 2 innate lymphoid cells. Disruption of neuroepithelial VIP–VIPR1 interactions resulted in increased susceptibility to invasive bacterial infection, which contrasted with enhanced resistance to parasite infection. Our work identifies a multi-tissue axis that controls type 1 and type 2 immunity, deciphering how neuroepithelial interactions distinctively set gut immunity programs.
神经系统与免疫系统协同调控黏膜屏障完整性。然而,肠道神经元是否通过建立神经上皮相互作用来协调免疫应答仍不清楚。该研究鉴定了差异调控肠道1型与2型免疫的神经上皮相互作用。肠道上皮细胞表达血管活性肠肽(VIP)受体1(VIPR1),对肠道VIP能神经元的化学遗传学调控可改变上皮来源的细胞因子。上皮细胞固有性Vipr1基因缺失导致1型免疫减弱,包括1型警报素减少和上皮内淋巴细胞数量下降。与此相反,上皮Vipr1缺陷则增强2型免疫,表现为2型警报素增加、簇细胞增多以及活化的2型固有淋巴细胞。破坏神经上皮VIP–VIPR1相互作用会增加侵袭性细菌感染的易感性,但对寄生虫感染却表现出增强的抵抗力。研究揭示了一个调控1型与2型免疫的多组织轴,阐明了神经上皮相互作用如何特异性地设定肠道免疫应答程序。
8. Age-dependent Zap70 expression in thymocytes regulates selection of the neonatal regulatory T cell repertoire
胸腺细胞中Zap70表达的年龄依赖性调控新生期调节性T细胞库的选择

马萨诸塞大学陈医学院病理学系、犹他大学医学院微生物学与免疫学
The Foxp3⁺ regulatory T (Treg) cell repertoire carries age-dependent biases, with neonatal subsets enriched for highly self-reactive clones. However, the thymocyte features distinguishing neonatal from adult Treg selection remain unclear. Here, we show that neonatal double-positive mouse thymocytes, unlike their adult counterparts, fail to upregulate Zap70 during thymic selection, creating a calcium signaling bottleneck. This attenuated Zap70-dependent signaling limits negative selection, allowing highly self-reactive clones to evade deletion. Modulating Zap70 expression alters this balance; reducing Zap70 in adults rescues development of these clones, whereas increasing Zap70 in neonates enforces their deletion. Similarly, enhancing neonatal calcium signaling via increased LAT Y136-mediated PLCγ1 activation promotes clonal deletion. Analysis of pediatric human thymi reveals that ZAP70 expression remains low during the first year of life, aligning with the peak window for thymic Treg cell development. These findings suggest that age-dependent Zap70 expression regulates negative selection and thymic Treg cell development.
Foxp3⁺调节性T(Treg)细胞库具有年龄依赖性偏好,其中新生期Treg亚群富含高自身反应性克隆。然而,区分新生期与成年期Treg细胞选择的胸腺细胞特征尚不清楚。该研究发现,与成年胸腺细胞不同,新生期小鼠双阳性胸腺细胞在胸腺选择过程中未能上调Zap70表达,从而形成钙信号传导瓶颈。这种减弱的Zap70依赖信号传导限制了阴性选择,使得高自身反应性克隆得以逃逸删除。调控Zap70表达可改变这一平衡:在成年期降低Zap70能挽救这些克隆的发育,而在新生期增加Zap70则强制其删除。同样,通过增强LAT Y136介导的PLCγ1活化来强化新生期钙信号传导也能促进克隆删除。对儿童人类胸腺的分析显示,ZAP70表达在出生后第一年内持续处于低水平,这与胸腺Treg细胞发育的高峰窗口期相符。这些发现表明,年龄依赖性的Zap70表达调控着阴性选择与胸腺Treg细胞的发育。
9. Pseudokinase STK40 limits T cell exhaustion through COP1-dependent degradation of AP1-family transcription factors
伪激酶STK40通过COP1依赖的AP-1家族转录因子降解途径延缓T细胞耗竭

美国Genentech生理化学部、计算科学部
Serine/threonine kinase 40 (STK40) belongs to the Tribbles family of pseudokinases, which serve as substrate adaptors for the CRL4COP1/DET1 E3 ubiquitin ligase complex. Tribbles-1 and Tribbles-2 promote the degradation of CCAAT/enhancer-binding protein (C/EBP) transcription factors in hematopoietic cells. STK40 also regulates C/EBP proteins, and although some immune system functions have recently emerged, its specific role in cytotoxic T cell responses has not been characterized. Here we show that murine STK40 restricts homeostatic and antigen-driven T cell expansion. During chronic viral infection, T cell-specific deletion of Stk40 improved viral clearance and reduced the proportion of terminally exhausted antigen-specific T cells. STK40-deficient T cells exhibited increased expression of markers of cell proliferation and AP-1 target genes. Biochemically, STK40 interacted with the AP-1 transcription factor c-Jun, and was required for its CRL4COP1/DET1-dependent proteasomal degradation. Collectively, our results identify the COP1/STK40 axis as a post-translational cell-intrinsic mechanism regulating T cell immunity.
丝氨酸/苏氨酸激酶40(STK40)属于Tribbles家族的伪激酶,该家族作为CRL4COP1/DET1 E3泛素连接酶复合物的底物衔接蛋白发挥作用。Tribbles-1和Tribbles-2可促进造血细胞中CCAAT/增强子结合蛋白(C/EBP)转录因子的降解。STK40同样调控C/EBP蛋白,尽管其部分免疫系统功能近期已被揭示,但它在细胞毒性T细胞应答中的具体作用尚未明确。该研究发现,小鼠STK40可限制稳态及抗原驱动的T细胞扩增。在慢性病毒感染过程中,T细胞特异性敲除Stk40基因能改善病毒清除并减少终末耗竭型抗原特异性T细胞的比例。STK40缺陷的T细胞表现出细胞增殖标志物及AP-1靶基因表达的增加。在生化机制上,STK40与AP-1转录因子c-Jun相互作用,并是其CRL4COP1/DET1依赖性蛋白酶体降解所必需的。总之,研究结果确定了COP1/STK40轴作为调控T细胞免疫的翻译后细胞固有机制。
10. Analysis of the three-dimensional genome of exhausted CD8+ T cells reveals a critical role of IRF8 in their differentiation and functions in cancer
对耗竭性CD8⁺ T细胞三维基因组的分析揭示IRF8在其分化及肿瘤免疫功能中的关键作用

清华大学免疫学研究所,西湖大学医学院,上海交通大学医学院附属仁济医院
CD8+ T cells are rendered exhausted in cancer, accompanied by extensive changes in the epigenome. However, whether and how higher-order chromatin organization is involved in exhausted CD8+ T cell differentiation is unclear. Here, we show extensive changes in the three-dimensional genome during T cell exhaustion associated with changes in gene transcription. Moreover, we identified interferon regulatory factor 8 (IRF8) as an essential transcription factor involved in reorganization of the spatial proximity between enhancers and promoters of genes associated with exhausted T cell differentiation. IRF8 deficiency inhibited the differentiation of exhausted CD8+ T cells and their antitumor function. Mechanistically, IRF8 bound to genes associated with exhausted T cell differentiation and promoted the formation of intra-TAD chromosomal loops. At the loop anchor regions, IRF8 recruited CTCF to form active chromosomal structures to regulate gene transcription. These results thus identify a critical role of IRF8-dependent chromatin topology during exhausted CD8+ T cell differentiation.
CD8⁺ T细胞在肿瘤微环境中发生耗竭,并伴随表观基因组的广泛改变。然而,高级染色质组织结构是否以及如何参与耗竭性CD8⁺ T细胞分化尚不明确。该研究发现,T细胞耗竭过程中三维基因组发生广泛改变,这些改变与基因转录变化相关。进一步,研究鉴定出干扰素调节因子8(IRF8)是参与重组耗竭性T细胞分化相关基因增强子与启动子空间邻近性的关键转录因子。IRF8缺失会抑制耗竭性CD8⁺ T细胞的分化及其抗肿瘤功能。机制上,IRF8结合于耗竭性T细胞分化相关基因,并促进拓扑关联结构域(TAD)内染色体环的形成。在染色体环锚定区域,IRF8招募CTCF以形成活性染色体结构来调控基因转录。因此,这些结果确定了IRF8依赖性染色质拓扑结构在耗竭性CD8⁺ T细胞分化中的关键作用。
11. Thrombospondin-1–CD47 signaling contributes to the development of T cell exhaustion in cancer
血小板反应蛋白-1–CD47信号传导促进肿瘤中T细胞耗竭的发生发展

威尔康奈尔医学院桑德拉与爱德华·迈耶癌症中心,旧金山帕克癌症免疫治疗研究所
T cell exhaustion is a major barrier to effective cancer immunotherapy. Although immune checkpoint blockade can reinvigorate exhausted T cells, not all patients achieve long-term responses, partly due to the refractory nature of terminally exhausted T cells. Beyond persistent antigen stimulation, the environmental drivers of exhaustion remain to be thoroughly characterized. Here we identify CD47 upregulation in tumor-infiltrating exhausted CD8+ T cells in both human and murine tumors. We reveal a novel role for the extracellular matrix protein thrombospondin-1 (TSP-1) in engaging CD47 on T cells to promote exhaustion. This interaction activates calcineurin–NFAT signaling, inducing upregulation of TOX and expression of inhibitory receptors, and impairing effector function during tumor progression. Importantly, disrupting the TSP-1–CD47 axis prevents T cell exhaustion and enhances tumor control. Our findings identify a novel pathway promoting T cell dysfunction and suggest that targeting the TSP-1–CD47 axis is a promising strategy to enhance T cell immunity and immunotherapy efficacy.
T细胞耗竭是有效癌症免疫治疗的主要障碍。尽管免疫检查点阻断能重振耗竭的T细胞,但并非所有患者都能获得长期应答,部分原因是终末耗竭T细胞的难治性。除持续性抗原刺激外,导致耗竭的环境驱动因素仍有待深入解析。该研究发现,在人类和小鼠肿瘤中,肿瘤浸润的耗竭性CD8⁺ T细胞均出现CD47上调。揭示了细胞外基质蛋白血小板反应蛋白-1(TSP-1)通过结合T细胞上的CD47促进耗竭的新功能。这种相互作用可激活钙调磷酸酶–NFAT信号通路,诱导TOX上调及抑制性受体表达,并在肿瘤进展过程中损害效应功能。重要的是,阻断TSP-1–CD47轴能预防T细胞耗竭并增强肿瘤控制。研究发现鉴定出一条促进T细胞功能失调的新通路,并提示靶向TSP-1–CD47轴是增强T细胞免疫及免疫治疗疗效的潜在策略。
12. Transcriptional regulator SATB1 limits CD8+ T cell population expansion and effector differentiation in chronic infection and cancer
转录调控因子SATB1限制慢性感染与肿瘤中CD8⁺ T细胞群体的扩增及效应分化

墨尔本大学彼得·多尔蒂感染与免疫研究所微生物学与免疫学系,波恩大学医院分子医学与实验免疫学研究所
CD8+ T cells are major mediators of antiviral and antitumor immunity. During persistent antigen stimulation as in chronic infection and cancer, however, they differentiate into exhausted T cells that display impaired functionality. Precursors of exhausted T (TPEX) cells exhibit stem-like properties, including high proliferative, self-renewal and developmental potential, and are responsible for long-term CD8+ T cell responses against persistent antigens. Here we identify the chromatin organizer and transcriptional regulator SATB1 as a major regulator of exhausted CD8+ T cell differentiation. SATB1 was specifically expressed in TPEX cells where it limited population expansion and effector differentiation while preserving functionality of CD8+ T cells. SATB1 downregulation was required for TPEX cell-to-effector cell differentiation in chronic infection and contributed to coordinated effector and memory differentiation in acute viral infection. DNA binding of SATB1 regulated gene expression both dependent and independent of chromatin accessibility. Finally, SATB1 limited antitumor CD8+ and chimeric antigen receptor T cell immunity. Overall, our results identify SATB1 as a central regulator of precursor fate and effector differentiation of CD8+ T cells both in infection and in cancer.
CD8⁺ T细胞是抗病毒与抗肿瘤免疫的主要效应细胞。然而,在慢性感染和肿瘤等持续性抗原刺激下,它们会分化为功能受损的耗竭性T细胞。耗竭前体T(TPEX)细胞具有干细胞样特性,包括高增殖、自我更新和发育潜能,负责针对持续性抗原的长期CD8⁺ T细胞应答。该研究发现,染色质组织因子兼转录调控因子SATB1是耗竭性CD8⁺ T细胞分化的关键调控因子。SATB1特异性表达于TPEX细胞中,其在限制细胞群体扩增与效应分化的同时,维持了CD8⁺ T细胞的功能完整性。SATB1下调是慢性感染中TPEX细胞向效应细胞分化所必需的,并有助于急性病毒感染中效应与记忆分化的协调进行。SATB1的DNA结合既能通过染色质可及性依赖途径,也能通过非依赖途径调控基因表达。最后,SATB1限制了抗肿瘤CD8⁺ T细胞及嵌合抗原受体T细胞的免疫应答。总体而言,研究结果确立了SATB1作为感染与肿瘤中CD8⁺ T细胞前体命运决定及效应分化的核心调控因子。

END
阅读最新文献,紧跟前沿进展,这是一名研究者必须具备的习惯和要求。我们华西医院耳鼻咽喉头颈外科的硕士、博士研究生和博士后们自2019年以来,每周开展一次文献泛读和文献精读分享会,至今已累计开展了200多次。2023年9月13日开始,本科室陆续将其进行整理,同步推出在线前沿速递和文献解读板块。通过这种学习和分享的方式,使汇报者和大家都能对近期权威期刊发表的高质量研究有所了解,同时也是学习其他优秀研究者思路、方法和理论的良好手段。希望通过这种形式,把科内的分享扩大到所有的读者,一起学习,共同进步!
华西医院耳鼻咽喉头颈外科

2023年9月13日



